In which of the following SN1 reaction is feasible?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Subtopic: Mechanism of Reactions | Chemical Properties |
82%
Level 1: 80%+
Please attempt this question first.
Hints
Please attempt this question first.
| Assertion (A): | Isopropyl chloride is more reactive than CH3Br in SN2 reactions. |
| Reason (R): | SN2 reactions are always accompanied by inversion of configuration. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Mechanism of Reactions | Chemical Properties |
81%
Level 1: 80%+
Please attempt this question first.
Hints
Please attempt this question first.
The given reaction will be the fastest in which of the following solvents?
CH3 CH2 CH2 Br + NaCN - CH3 CH2 CH2 CN + NaBr
1. Ethanol
2. Methanol
3. N, N'-dimethylformamide (DMF)
4. Water
Subtopic: Mechanism of Reactions | Chemical Properties |
81%
Level 1: 80%+
NEET - 2016
Hints
Match the column:
1. P-IV, Q-III, R-II, S-I
2. P-I, Q-II, R-III, S-IV
3. P-II, Q-III, R-I, S-IV
4. P-III, Q-II, R-I, S-IV
| Column-I (Name reaction) |
Column -I (Representation of reaction) |
||
| (P) | Finkelstein reaction | (I) |  |
| (Q) | Lucas reaction | (II) |  |
| (R) | Wurtz reaction | (III) |  |
| (S) | Gattermann-Koch reaction | (IV) |  |
1. P-IV, Q-III, R-II, S-I
2. P-I, Q-II, R-III, S-IV
3. P-II, Q-III, R-I, S-IV
4. P-III, Q-II, R-I, S-IV
Subtopic: Mechanism of Reactions |
95%
Level 1: 80%+
Please attempt this question first.
Hints
Please attempt this question first.
The most reactive compound for SN2 reaction among the following is:
| 1. | 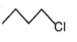 |
2. | 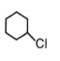 |
| 3. | 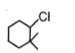 |
4. | 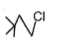 |
Subtopic: Mechanism of Reactions |
90%
Level 1: 80%+
Please attempt this question first.
Hints
Please attempt this question first.
In the following pairs of halogen compounds, which compound undergoes faster SN1 reaction?

The correct option is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Subtopic: Mechanism of Reactions |
89%
Level 1: 80%+
Hints
Choose the incorrect statement regarding SN2 reactions:
| 1. | As the concentration of nucleophiles increases, the reaction rate increases. |
| 2. | Both the attack of the nucleophile and the departure of the leaving group occur simultaneously. |
| 3. | Inversion of configuration occurs on asymmetric centers because the nucleophile attacks from the backside of the leaving group. |
| 4. | The SN2 mechanism is predominant in tertiary alkyl halides. |
Subtopic: Mechanism of Reactions |
87%
Level 1: 80%+
Please attempt this question first.
Hints
Please attempt this question first.
Identify the compound Y in the following reaction.
1.
2.
3.
4.
Subtopic: Mechanism of Reactions |
87%
Level 1: 80%+
Please attempt this question first.
Hints
The major product C in the below-mentioned reaction is:


| 1. | Propan-1-ol | 2. | Propan-2-ol |
| 3. | Propane | 4. | Propyne |
Subtopic: Mechanism of Reactions |
82%
Level 1: 80%+
NEET - 2024
Hints





 Product of reaction is :
Product of reaction is :




